
13C and 1H chemical shift assignment reported in Supplementary tables S1- S3 were determined on 20 mM samples. Samples were at concentration ranging from 1 μM to 20 mM. 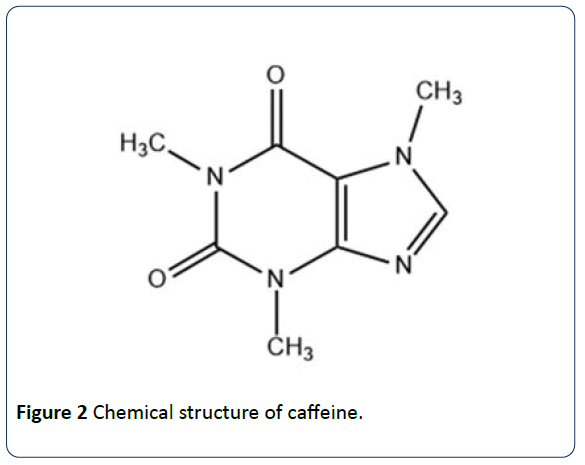
Caffeine and polyphenols were dissolved in H 2O (10 % D 2O) containing TSP (sodium 3-(trimethyl-silyl) propionate-2,2,3,3, d4) as internal standard for chemical shift referencing. Solutions containing polyphenols were prepared in 80 mM phosphate buffer at pH 7 in order to prevent pH effect on chemical shift at different concentrations. 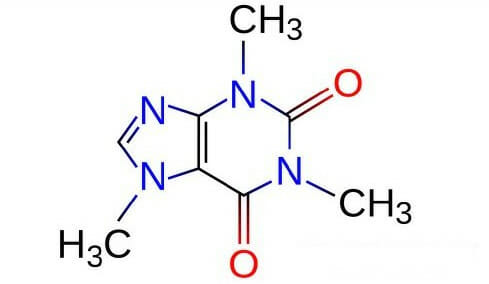
Louis, MO) and used without further purification. Caffeine and all other chemicals were purchased from Sigma Aldrich (Sigma Chemical Corp., St. ģ,5-di-O-caffeoylquinic, 3,4-di-O-caffeoylquinic and 4,5-di-O-caffeoylquinic acids were purchased from Phytolab GmbH (Germany). In particular, in addition to 5-O-caffeoylquinic acid, many other trans-cinnamic (hydroxycinnamic) acids esters with (-)-quinic acid (Scheme 1, according to IUPAC: 1 L-1 (OH),3,4/5-tetrahydroxycyclohexanecarboxylic acid ( 1) name revised in 1997 as 1s n,3R,4s n,5R-tetrahydroxycyclohexanecarboxylic acid ), are present in coffee brews. The present investigation has been largely stimulated by the hypothesis that other coffee polyphenols could be involved in caffeine binding. 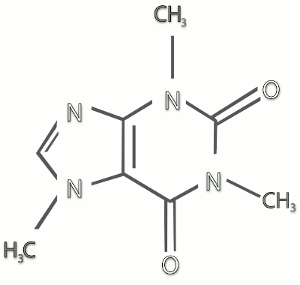
In espresso coffee, this molar fraction appears lower than expected, suggesting that other species may be involved in the binding of caffeine. Based on the equilibrium constant and the concentration of both complex constituents in water solution, the theoretical molar fraction of bound caffeine can be estimated. The complex has also been studied in freshly prepared espresso coffee brews. The caffeine-chlorogenate complex has been re-investigated by high resolution 1H-NMR spectroscopy and its structure has been revised. Recently, it has been proposed as a possible strategy to develop an analytical method to determine caffeine by means of capillary electrophoresis. The caffeine-chlorogenate complexation has been indicated as crucial mechanism to explain the compartmentation of caffeine in Coffea plants and the qualitative relationship between caffeine and chlorogenic acid content distribution among wild Coffea species. In the 70’s, Horman and Viani, on the basis of NMR chemical shift data, proposed that the caffeine-chlorogenate complex might be described as a 1:1 type π-molecular complex stabilized by hydrophobic interations. to study caffeine as well as other purine alkaloids complexation with chlorogenate. The same method has been used by Kappeler et al. determined the equilibrium constant of the complex formation reaction in water by spectrophotometry. The complex formed between caffeine and chlorogenic acid (5-O-caffeoylquinic acid according to IUPAC nomenclature, ), the well known major polyphenolic constituent of green coffee bean, was isolated one century ago by Gorter. Our results show that stacking interactions drive the formation of the complexes and that multiple equilibria are present in the interaction of caffeine with 3,4-di-O-caffeoylquinic acid and 4,5-di-O-caffeoylquinic acid while the complex with 3,5-di-O-caffeoylquinic acid seems to be better defined. Structural features of the three different complexes have also been investigated by NMR spectroscopy combined with quantum chemical calculations, and the complex conformation is discussed. The value of the association constant of the caffeine-di-O-caffeoylquinic acid complexes is compatible with previous studies and within the typical range of reported association constants for other caffeine-polyphenols complexes. Caffeine-di-O-caffeoylquinic acid isomers association constants were measured. Self-association of the di-O-caffeoylquinic acid isomers has been studied as well. Caffeine hetero-association with 3,5-di-O-caffeoylquinic acid, 3,4-di-O-caffeoylquinic acid and 4,5-di-O-caffeoylquinic acid in aqueous solution has been investigated by one-dimensional (1D) and two-dimensional (2D) high resolution 1H and 13C NMR spectroscopy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
